AI Medical Service Inc. Announces Regulatory Approval of Gastric AI-based Endoscopic Diagnostic Imaging Support System
- The endoscopic AI detects lesions suspicious for early gastric cancer -
AI MEDICAL SERVICE INC. (hereinafter AIM), a medical start-up specializing in the development of diagnostic endoscopic AI, is excited to announce the manufacturing and marketing approval for an AI-based endoscopic diagnostic support device that operates within the stomach (brand name: gastroAI-model G, registration number: 30500BZX00297000). The regulatory approval was granted by Japan’s Minister of Health, Labour and Welfare on December 26, 2023. The endoscopic AI (hereinafter gastric cancer diagnosis support AI) assists the physician in diagnosis by detecting lesion candidates for biopsy[1] or other additional diagnostic steps based on gross visual features during endoscopy. The product is scheduled to go on sale around March 2024.
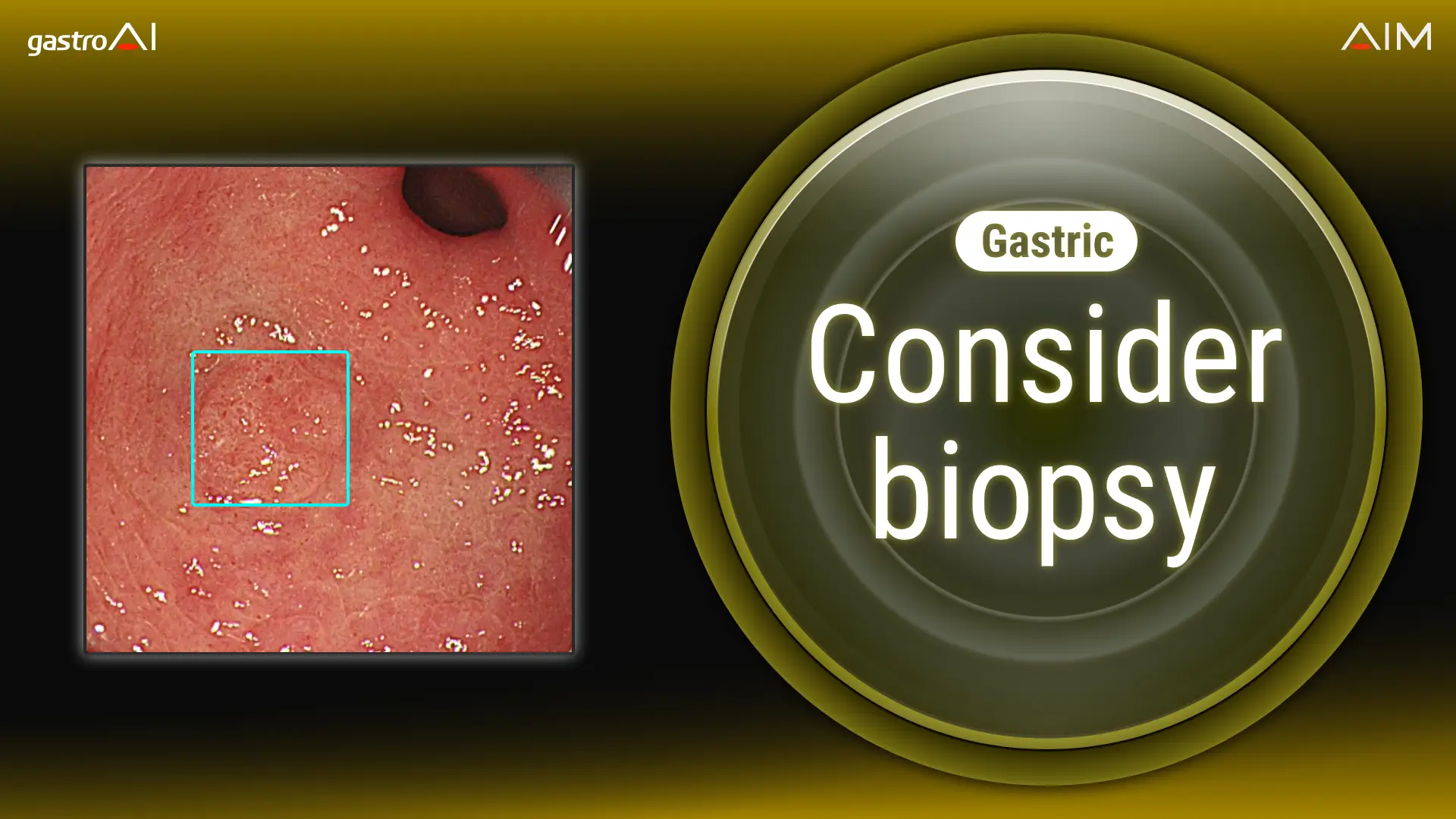
About gastric cancer diagnosis support AI gastroAI-model G
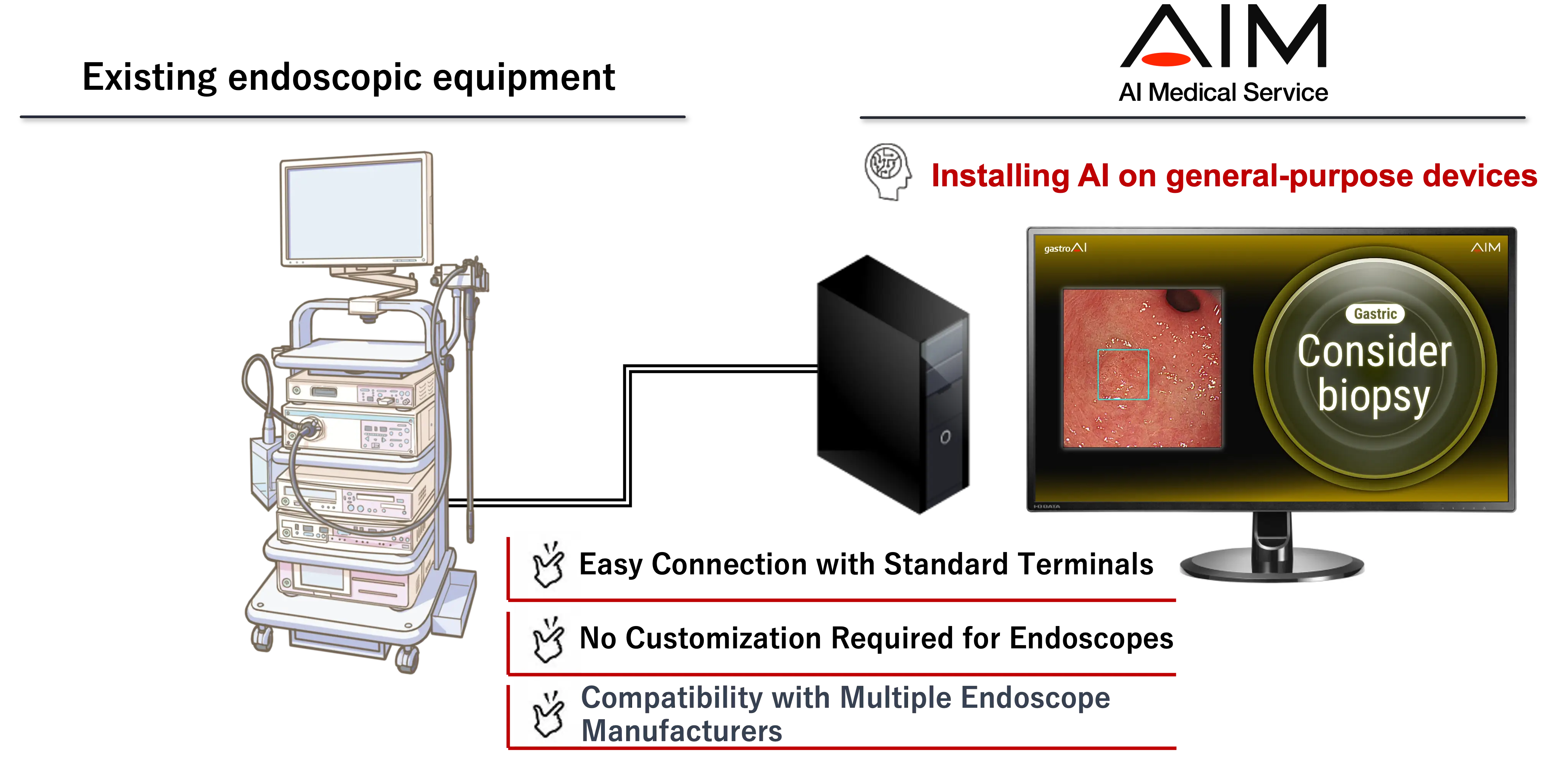
The gastric cancer diagnosis support AI, brand-named gastroAI-model G, estimates whether the lesion is a candidate for biopsy or other additional examination based on its gross visual characteristics. The AI works by scanning for candidate lesions using a general-purpose computer, using images gathered from the endoscopy system’s video processor. When the program calculates that a lesion is suspicious for early gastric cancer and warrants further workup, it alerts the physician and provides diagnostic assistance by displaying a rectangle inside the endoscopic display area.
The software was developed based on training data provided by world-class medical institutions, utilizing deep learning technology and the support of more than 100 collaborative research institutes.
The application was initially submitted for approval as “AI to differentiate between the neoplastic or non-neoplastic nature of gastric lesions,” but following discussions with the PMDA, the software has now been approved for “detection support use.”[2]
Brand name | Endoscopic Diagnosis Support Software gastroAI-model G |
Registration number | 30500BZX00297000 |
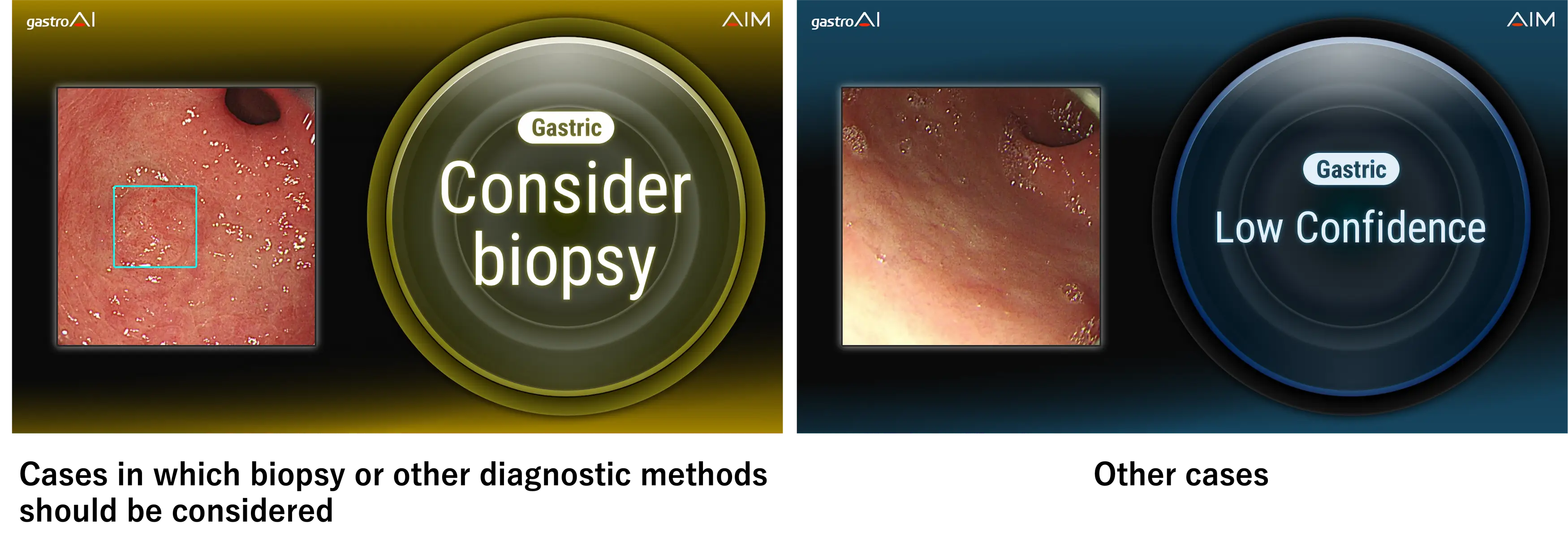
Main Features
1. Detection support by AI trained on a large amount of high-quality data
AI that facilities lesion detection after having been trained on numerous images provided by world-class medical institutions. This AI was trained on many lesion images containing early gastric cancer.
2. Display of analysis results during endoscopy
When a suspicious lesion is first seen by the physician and the freeze operation is performed, the AI begins its analysis and displays the results on the monitor.
3. Can be used with major endoscope manufacturers
This product can be connected to endoscope systems manufactured by Olympus or Fujifilm.
For inquiries about AIM’s products, please contact us through the following:
https://www.ai-ms.com/contact
Background of our efforts to obtain the regulatory approval
In Japan, the number of new gastric cancer cases each year is third only to colorectal cancer and lung cancer, with more than 130,000 cases each year; the number of deaths is the third most after colorectal cancer and lung cancer, with more than 40,000 deaths each year[3]. Gastric cancer is characterized by a mortality rate that increases significantly with disease progression. Furthermore, gastric cancer is very often treatable if detected at an early stage; the 5-year relative survival rate is approximately 95% if detected at stage I, but less than 50% if detected at stage III or later. Nevertheless, early-stage gastric cancer is difficult to detect and is said to be missed in 4.5 to 25.8% of cases[4].
Considering these circumstances, AIM has developed the gastric cancer diagnosis support AI. Utilizing the gastric cancer diagnosis support AI, it is possible to detect early-stage gastric cancer with the same level of accuracy as a specialist. It is expected that use of the AI will reduce the number of missed early-stage gastric cancers and equalize the quality of endoscopic examinations.
The gastric cancer diagnosis support AI offers advantages for both patients and physicians. For patients, it is expected to improve the accuracy of endoscopic examinations through a synergy that exists between physicians and the gastric cancer diagnosis support AI during routine clinical activities. For the physician, use of the AI can reduce the number of overlooked cancer cases and somewhat alleviate the psychological burdens of medical decision-making, all without the need for learning a complicated new technique.
Furthermore, when gastric cancer is detected at an early stage, "endoscopic treatment," in many cases, it is possible to resect lesions from the mucosa using an endoscope. Compared to surgical procedures like partial or total gastric resection, endoscopic lesion resection places relatively few physical and financial burdens on the patient while leading to superior prognosis and quality of life.
AIM will continue to strive to realize its mission of "Save Lives All Over the World" through endoscopic image diagnosis support AI.
Future Developments
With the mission of “Save Lives All Over the World,” AIM will deliver endoscopic AI (including but not limited to gastric cancer diagnosis support) to clinical sites not only in Japan but also around the world. Japanese manufacturers lead the world in the development and manufacturing of gastrointestinal endoscopes, and Japanese outfits have accumulated the highest quality and largest volume of imaging data. In the future, AIM will accelerate R&D to expand the functions of gastrointestinal diagnosis support AI and to expand the target organs to solve problems in clinical practice.
By developing an endoscopic AI that draws on the wisdom of Japanese endoscopists, we hope to improve endoscopic care in Japan and around the world in order to save as many patients as possible.
About AI Medical Service Inc.
AI Medical Service (AIM) is a Tokyo-based med-tech company established with the mission to “Save Lives All Over the World.” Japan leads the world in endoscopic diagnosis and treatment, providing firms and researchers with access to large amounts of high-quality data. AIM is the leading player in the field of endoscopic AI, engaging in joint research with more than 100 medical institutions. By bringing endoscopic AI to the real-world clinical setting as soon as possible, AIM aims to reduce the number of missed cancer diagnoses and save lives around the world.
About CEO of AIM, Dr. Tomohiro Tada

Tomohiro TADA, M.D., Ph.D.
Dr. Tomohiro TADA is the CEO of AI Medical Service Inc., the chairperson of Tada Tomohiro The Institute of Gastroenterology & Proctology as well as Visiting Lecturer, Department of Surgical Oncology, Graduate School of Medicine, the University of Tokyo Hospital.
Dr. TADA received his M.D. of school of medicine in 1996 and Ph.D. of department of surgery in 2005 from the University of Tokyo. He trained in Colorectal Surgery at the University of Tokyo Hospital.
―――――――――――――――――――――――――――――
Company: AI Medical Service Inc.
Address: Hareza Tower 11F, 1-18-1 Higashiikebukuro, Toshima-ku, Tokyo 170-0013, Japan
CEO: Tomohiro Tada
Founding: September 1, 2017
Business: Development of Endoscopic AI
[1] “Biopsy” refers to taking a piece of tissue and examining it under a microscope when a lesion is found.
[2] In the pharmaceutical application process, additional studies were conducted due to a change in the intended use of the product, and the product satisfied the criteria required by the PMDA for detection use.
[3] Reference: Cancer Today [Accessed 2023/10/17]
https://gco.iarc.fr/today/
[4] Reference: Hosokawa et al. Hepatogastroenterology 2007 Mar;54(74):442-4.
Contact
Feel free to contact us using the form below regarding joint research,
media coverage, business partnerships, or related opportunities.