AI Medical Service Inc. receives manufacturing and marketing approval in Japan for Endoscopic Image Diagnosis Support Software “gastroAI model-G3”
AI MEDICAL SERVICE INC. (hereinafter AIM) is pleased to announce that it has obtained manufacturing and marketing approval for “gastroAI model-G3,” an AI-based endoscopic lesion detection support software for Upper GI endoscopy. The approval was granted on May 13, 2026. This endoscopic AI (hereinafter referred to as the “gastroAI model-G3”) analyzes gastric endoscopic videos acquired by physicians for lesion detection and identifies regions suspicious for early gastric cancer and adenoma[1] in real-time. “gastroAI model-G3” will highlighting the suspicious regions to support physicians observation and lesion detection[2]. The product will be launched in phases beginning in the summer 2026 in Japan.

About gastric cancer detection support AI “gastroAI model-G3”
“gastroAI model-G3” will support physician’s observation by highlighting suspicious lesions when detected. “gastroAI model-G3” overlays up to three markers indicating potentially suspicious regions such as early gastric cancers and adenomas in real-time.
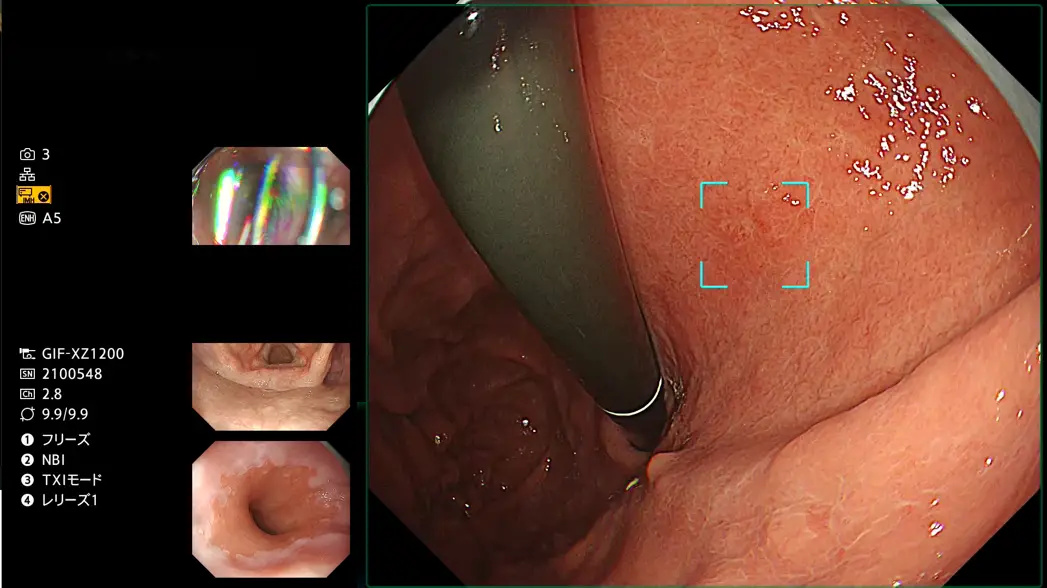
Brand name:Endoscopic image diagnosis support software AP01
Generic Name:Endoscopic Image Diagnosis Support Program for Lesion Detection
Registration number:30800BZX00135000
Detection Performance
By expanding the diversity of the training data, optimizing the AI model, and refining the decision-making process through multi-frame analysis, the software achieves both high detection performance and reduced false positives. In addition, the software is equipped with an automatic recognition function that detects images unsuitable for analysis and temporarily suspends analysis when necessary. These features help reduce excessive detections and support more efficient diagnosis by enabling physicians to focus on regions that truly require further examination.
【Performance Evaluation Test Results】
Description: Retrospective performance evaluation study[3] using gastric endoscopic videos and pathological information.
Subjects: Lesions diagnosed as early gastric cancer (raised, flat, or depressed) and adenoma.
Number of samples: 1,099.
Sensitivity(%) | 94.6 |
Specificity(%) | 94.3 |
Background of our efforts to obtain regulatory approval
In Japan, gastric cancer continues to rank among the leading cancers in both incidence and mortality. Approximately 100,000 people are newly diagnosed with gastric cancer each year, making it the third most common cancer after colorectal cancer and lung cancer, while the number of deaths ranks fourth among all cancers[5]. Gastric cancer is considered a highly treatable disease when detected at an early stage; however, survival rates decline significantly as the disease progresses. The 5-year relative survival rate exceeds 95% when gastric cancer is detected at Stage I, but falls below 50% when detected at Stage III or later. Early detection is therefore critically important, yet early-stage gastric cancer remains difficult to identify, with studies suggesting that approximately 4.5% to 25.8% of cases may be missed[6].
With the newly approved “gastroAI model-G3”, real-time analysis of endoscopic video during endoscopic examination has become possible. This enables the AI to continuously assist physicians throughout the entire examination process, from start to finish. Against the backdrop of challenges such as regional disparities in physician availability, “gastroAI model-G3” is intended for approximately 18,000 medical institutions[7] across Japan that perform endoscopic examinations. AIM expects further business expansion, including enhanced value delivery to existing customer sites and medical institutions already using its products.
By delivering endoscopic AI to the real-world clinical setting as soon as possible, AIM aims to support lesion detection during endoscopic examination and save lives around the world.
About AI Medical Service Inc.
AI Medical Service is a med-tech company and healthcare startup guided by the mission to "Save Lives All Over the World." Japan is a world leader in the field of endoscopy, with an accumulation of world-class data in both quality and quantity. We are engaged in product research and development that leverages this data, and we aim to save patients worldwide by swiftly delivering the results of these efforts to clinical settings.
[1]Benign tumor. Large lesions may be treated endoscopically because they can become cancerous.
[2]The product is positioned as an “examiner’s aid in detecting lesions” and is not intended to support qualitative diagnosis or determine a treatment strategy based solely on the results of analysis using the product.
[3]Study using anonymized medical data (removed medical records number, etc.) from past medical procedures.
[4]The lower bound statistically estimated with 97.5% confidence that the true value is equal to or greater than this value.
[5]Reference: Cancer Information Service, National Cancer Center Japan, “Cancer Statistics” (National Cancer Registry).
[6]Reference:Hosokawa et al. Hepatogastroenterology 2007 Mar;54(74):442-4.
[7]Reference: Ministry of Health, Labour and Welfare, “Survey of Medical Institutions” (e-Stat).
Contact
Feel free to contact us using the form below regarding joint research,
media coverage, business partnerships, or related opportunities.